Updated April 2025
On this page
What is food allergy?
A food allergy is an immune system response, almost always to a food protein, that the body mistakenly believes is harmful.
When a person eats food containing the allergen, the immune system releases a large amount of chemicals, including histamine, that lead to signs and symptoms of an allergic reaction.
An allergic reaction can involve the gut, skin and breathing and/or heart systems. Symptoms of food allergy can be mild to moderate or severe (known as anaphylaxis).
Anaphylaxis is a life-threatening emergency. Hospital presentations for anaphylaxis have increased for all age groups in the past 30 years with food being the major cause of anaphylaxis in the 0-4 age group.
Triggers and prevalence
The most common allergens are cow’s milk (dairy), egg, peanut, tree nuts, sesame, fish, crustacean, mollusc, wheat and soy. As well as these common allergens, lupin must also be listed on a packaged food label, no matter the amount.
Many other foods including kiwi fruit, banana, chicken, mustard and celery can also cause allergic reactions, including anaphylaxis. There are more than 170 foods known to have triggered allergic reactions.
In Australia, food allergy is common and estimated to affect around 1 in 10 babies, 1 in 20 teens and 1 in 50 adults.

Most children, not all, will grow out of their allergy to cow’s milk, egg, soy and wheat. However, allergies to peanut, tree nuts, sesame and seafood tend to be lifelong.
If a person develops a food allergy as an adult, they are likely to have that allergy for the rest of their lives.

Babies and food allergies
When to introduce food to babies, allergic reactions in babies and more.
Food Allergens
It is important to understand that even very small amounts of a food or drink can lead to anaphylaxis in some people. A&AA has developed food allergen cards for each of the common food allergens as well as Mammalian Meat Allergy. The food allergen cards list foods and ingredients that the food allergen is commonly found in.
You can learn more about the most common food allergens below.
Other food allergens
More than 170 food are known to have triggered allergic reactions.
Food allergy or intolerance?
Knowing the difference between a food allergy and a food intolerance can be confusing for many people as some of the signs and symptoms of a mild to moderate food allergy are similar to the signs and symptoms of a food intolerance. Unlike a food allergy, a food intolerance does not involve the body’s immune system and is not life-threatening.
Read more about the difference between a food allergy and intolerance.
Signs & Symptoms of a food allergic reaction
Signs and symptoms of a food allergy usually occur soon after eating the food or within two hours of eating. Allergic reactions to food can be mild, moderate or severe.
An allergic reaction may start with mild or moderate signs or symptoms but can progress rapidly to anaphylaxis. However, it is important to remember that signs of a mild or moderate allergic reaction may not always occur before anaphylaxis.
Signs and symptoms of an allergic reaction
Learn about the signs and symptoms of allergic reactions, and the difference between a mild or moderate reaction and a severe reaction (anaphylaxis).
Food allergy management
1. Know how to reduce the risk of an allergic reaction:
- Know what is in your food and drink.
- Read food labels. Look for your allergen in the ingredients list, and for precautionary allergen labelling on packaging. Check ingredients each time you buy the food because they can change.
- Always tell food service staff about your allergy and ask about ingredients, preparation and cross contamination risks. Don’t rely on menu descriptions when ordering foods and drinks at restaurants. Give staff a chef card which has the foods you are allergic to written on it to make it easier for the chef.
- Be aware your food allergen may be hidden in foods and drinks, including alcohol.
- Wash hands before eating.
- Learn how to store and prepare food.
- Cosmetics may contain food allergens so check ingredients. Requirements for food allergen labelling on cosmetics are not as strict as labelling laws on foods. Cosmetics do not have to have clear labels like foods do.
2. Know how to
treat an
allergic reaction:
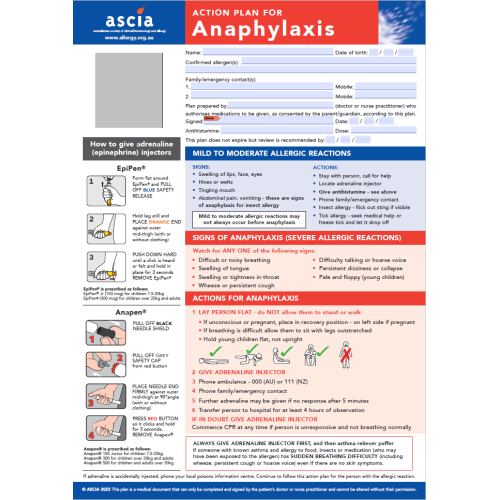
- Always have at least 1 adrenaline device with you (if prescribed), as well as your ASCIA Action Plan.
- Know the signs of an allergic reaction. Use the ASCIA action plan to help you.
- Follow the instructions on your ASCIA Action Plan if you have an allergic reaction.
- Be prepared for an emergency – educate yourself so you will know what to do.
- Emergency action – how to treat anaphylaxis.
3. Know how to manage your food allergy in the following settings:
Never eat or drink unless you have your adrenaline device(s) with you.
When to seek help
If you think that you or a family member has had an allergic reaction to a food, it is important to that you make an appointment to speak with your General Practitioner (GP). If your GP believes it is food allergy, they should refer you to a clinical immunology/allergy specialist. The Australasian Society of Clinical Immunology and Allergy (ASCIA) has a list of specialists in each state.
Treatment
There is no cure for food allergy. Currently, there are different ways to manage food allergies. Most people with food allergy must avoid foods that cause their allergic reaction although some people may be able to eat small amounts of the food if it is cooked, such as egg or milk in baked products.
As researchers learn more about food allergies other options are being developed.
Oral Immunotherapy (OIT) is a special treatment program sometimes offered and supervised by allergy specialists. There are also new drugs being used to help manage the risk and severity of allergic reactions to food.
With all these options there is still the chance of an allergic reaction, including anaphylaxis, so people need to be informed and prepared.
Severity of allergic reaction
Deaths from food allergy are rare in Australia. We do not know the exact numbers because currently there is no register that collects this information. Allergy & Anaphylaxis Australia (A&AA) has learned from coroners’ inquests and our contact with families who have lost loved ones to food anaphylaxis that deaths from food anaphylaxis are almost always preventable.
The most common cause of death from anaphylaxis is when adrenaline has not been administered or there has been a delay in the administration.
It’s important to remember that most people who experience anaphylaxis recover from it. Fatal anaphylaxis is most commonly caused by medication or insect stings, particularly in adult males over the age of 50 years with other health conditions. However, there are recorded deaths from food anaphylaxis in Australia. Death from anaphylaxis remains rare.
People can experience their first severe allergic reaction at any age.
A number of factors can affect the severity of an allergic reaction to food:
- The severity of the allergy
- The amount of food eaten
- Exercise around the time the food was eaten
- Posture during the allergic reaction (standing or walking when having anaphylaxis)
- Asthma (whether it is well controlled)
- Alcohol at the time the food is eaten
- The way the food is prepared such as raw versus cooked, fatty meal, liquid
- Being unwell at the time the food is eaten
- Menstrual cycle (having your period at the time the food was eaten)
Read more about factors that affect an allergic reaction.
Diagnosis and testing
Related information
Living with food allergy
Learn more about daily life with food allergies – including reading labels and ingredients and planning ahead for eating in different environments.
Emergency preparation
Learn more about preparing for an emergency, and how to identify and respond to an allergic reaction including anaphylaxis.